A complex phenomenon
Autoimmune diseases form very broad spectrum encompassing a diversified range of disorders that vary in terms of disease severity and site of organic damage.
As such, autoimmunity includes skin disorders such as atopic dermatitis or psoriasis, forms of chronic pain such as rheumatoid arthritis, rheumatic polymyalgia, fibromyalgia, Chronic Fatigue Syndrome (CFS), airway diseases due to allergy and inflammation (allergic rhinitis, asthma), atherosclerosis, multiple sclerosis, allergic conjunctivitis. Still, despite these clear differences, all autoimmune diseases feature chronic inflammation [9,11,16,18,19,21,31,51,58] coupled with neuroendocrine alteration that prevents the extinction of the inflammatory hotspot. All of the above-mentioned diseases show increased expression of proinflammatory cytokines such as IL-2, IL-6 and TNF-α [8,60] and inflammatory markers (such as the C-reactive protein) [15].
Studies consistently report a deficit in the immunoendocrine axis response in patients with these diseases and a resulting difficulty or impossibility in curbing the inflammatory activity underway. [1,10,15,26-28,31,41,43-45,49,51,53,56,59,62]
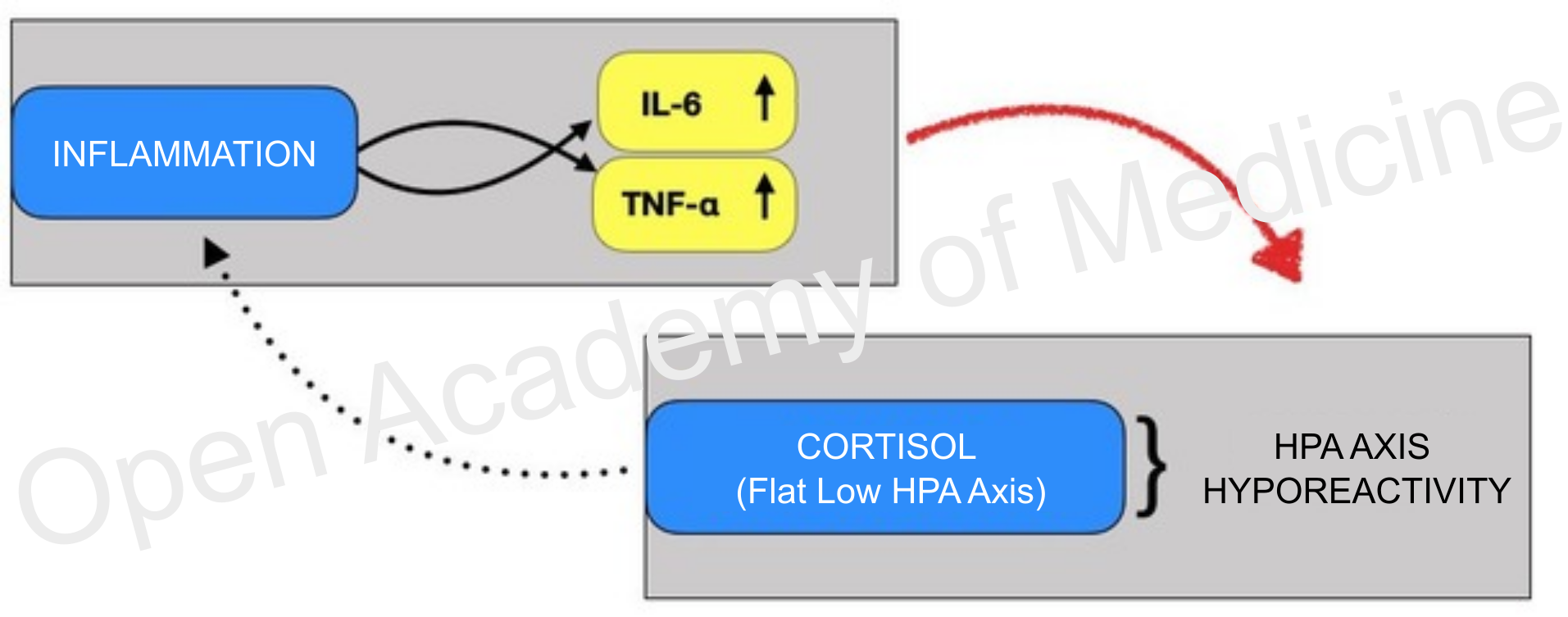
![]()
Figure 1: the immunoendocrine axis response is unable to mitigate inflammation
Cortisol and ACTH baseline levels do not seem to significantly differ between patients and controls. Instead, cortisol/CRP and ACTH/CRP ratios have been found to collapse. In addition, the stimulation test done with ACTH produces a significantly mitigated response in terms of cortisol release. [5,24,36,47]
A hallmark of autoimmunity is a flat low HPA axis (HPA axis index: Flat Low, BIA-ACC) or in any case a flat low adrenal cortex (this being the ultimate trigger of the HPA axis). In addition to poor secretion of cortisol in response to stressors, a degree of resistance or inertia that slows down the restoration of baseline cortisol levels has also been found. The latter plays a role in the disruption of the circadian rhythm of the neuro-immunoendocrine axis and frequent arrhythmic lowering and flattening of baseline plasma cortisol levels, often worsened by thyroid function inhibition (hypophysis lowering of TSH and GH). [12,22,29,30,37,40,54]
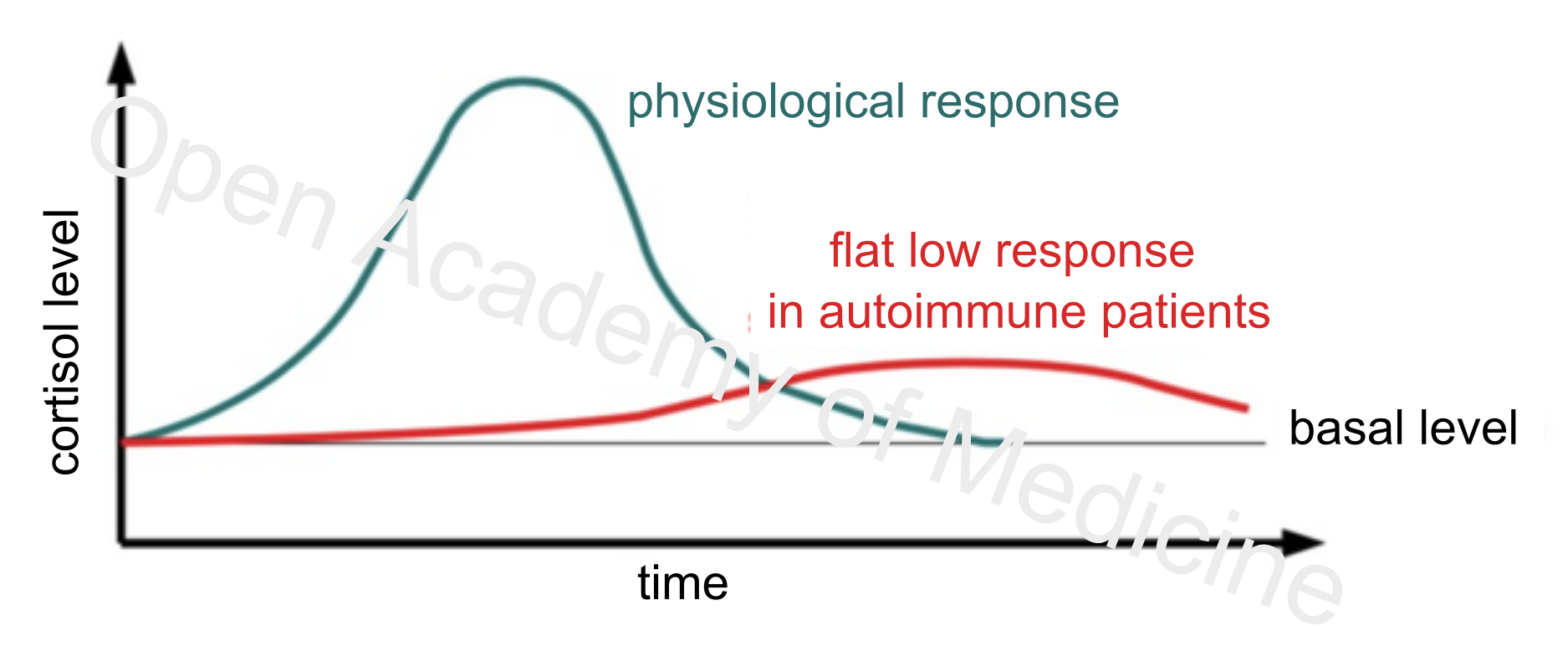
![]()
Figure 2: response to a stressor in a healthy individual as compared to one with autoimmunity
Still, it seems that the alteration of the HPA axis and its interactions with the sympathetic nervous system (PPG Stress Flow) cannot be properly described by generically labelling them as being flat low. Indeed, with the hypothalamus and the release of CRH, the situation can be the opposite, with excess (proinflammatory) central and peripheral CRH [6,35,48,52,63] that, in turn, correlates to decreased TSH and GH. Other experiences provide information on the increase in baseline noradrenaline (due to overstimulation of the Sympathetic Nervous System - SNS) and the response of noradrenaline to inflammatory stimuli (in particular to increased IL-6) in patients with fibromyalgia [2,3,38,50], which highlights an altered regulation of the sympathetic nervous system (post-stress increase in the heart rate, inhibited insulin secretion, altered thermoregulation, etc.).
Autoimmunity-related issues:
Recovery strategies
As such, whatever the cause, autoimmune diseases are triggered by inflammation, the inability of the neuro-immunoendocrine reaction to mitigate and eventually extinguish inflammation, resulting in damage to healthy tissues.
Clearly, it is necessary to limit the damage produced by chronic inflammation. It is therefore fundamental to take into consideration the degree of systemic inflammation (that can be evaluated through: ECW extracellular water - BIA-ACC, RMSSD vagal activity and capillary permeability - PPG Stress Flow) and to adopt strategies that are suitable to progressively curb it so as to reduce the incidence, or at least the intensity, of symptoms.
By integrating very useful information, it is possible to swiftly restore major biological rhythms lost due to chronic inflammation.
The main goal is to create rhythmicity, in biological, physical and metabolic terms, between the acrophase and the batiphase. The dominance of the sympathetic nervous system (LF range – sympathetic nervous system – PPG Stress Flow) during the day and the dominance of the parasympathetic nervous system (HF range – parasympathetic nervous system – PPG Stress Flow) in the evening enables to ensure this rhythmicity.
Besides, adding, as appropriate, buffer systems supplements is useful to counter the subclinical acidosis that is typically associated with chronic inflammation.
A negative PRAL diet that takes into account the proper glycemic load (GL) between day and night, is equally useful to counter subclinical the acidosis associated to inflammatory load, while strongly supporting the acrophase (nervous system and cortisol in synch).
In view of mitigating inflammation, besides stimulating the immunoendocrine system, one should consider taking omega-3 poly-unsaturated fatty acids: EPA and DHA are particularly indicated for suppressing the inflammatory processes correlated to autoimmune diseases, mainly by inhibiting the synthesis of arachidonic acid derived proinflammatory eicosanoids (omega-6). [13,20,23,25,33-34,39,42,46,61]. As a matter of fact, many have demonstrated a decrease in the main inflammatory markers (in particular IL-6 and TNF-α) as a result of taking these omega-3 supplements. [23,66]
Recent studies [7,14,17,64] have shown the anti-inflammatory properties of hops extracts (Humulus lupulus L.) as proven by lower CRP, particularly in treating chronic pain such as rheumatoid arthritis and other diseases associated to chronic inflammation coupled with chronic stress.
Authors: Dario Boschiero, Mariantonietta Lucafò - Date: 22/02/2021
Attention: these contents can be freely used for personal learning purposes only. The use is regulated by Law No. 633/1941 and subsequent amendments, as well as by the copyright and patent legislation in force. Any use for commercial and profit-making purposes is forbidden.
References
- Sulli A, Montecucco CM, Caporali R, Cavagna L, Montagna P, Capellino S, Fazzuoli L, Seriolo B, Alessandro C, Secchi ME, Cutolo M. Glucocorticoid effects on adrenal steroids and cytokine responsiveness in polymyalgia rheumatica and elderly onset rheumatoid arthritis. Ann N Y Acad Sci. 1069:307-14; 2006
- Sarzi-Puttini P, Atzeni F, Diana A, Doria A, Furlan R. Increased neural sympathetic activation in fibromyalgia syndrome. Ann N Y Acad Sci.1069:109-17; 2006
- Geenen R, Van Middendorp H, Bijlsma JW. The impact of stressors on health status and hypothalamic-pituitary-adrenal axis and autonomic nervous system responsiveness in rheumatoid arthritis. Ann N Y Acad Sci.1069:77-97; 2006
- Harbuz MS, Richards LJ, Chover-Gonzalez AJ, Marti-Sistac O, Jessop DS. Stress in autoimmune disease models. Ann N Y Acad Sci.;1069:51-61; 2006
- Demir H, Tanriverdi F, Ozogul N, Calis M, Kirnap M, Durak AC, Kelestimur F. Evaluation of the hypothalamic-pituitary-adrenal axis in untreated patients with polymyalgia rheumatica and healthy controls. Scand J Rheumatol. 35(3):217-23; 2006
- O'Kane M, Murphy EP, Kirby B. The role of corticotropin-releasing hormone in immune-mediated cutaneous inflammatory disease. Exp Dermatol.15(3):143-53; 2006
- Hougee S, Faber J, Sanders A, Berg WB, Garssen J, Smit HF, Hoijer MA. Selective inhibition of COX-2 by a standardized CO2 extract of Humulus lupulus in vitro and its activity in a mouse model of zymosan-induced arthritis. Planta Med. 72(3):228-33; 2006
- Gabay C. Interleukin-6 and chronic inflammation. Arthritis Res Ther. 8 Suppl 2:S3. 2006
- Hamminga EA, van der Lely AJ, Neumann HA, Thio HB. Chronic inflammation in psoriasis and obesity: implications for therapy. Med Hypotheses. 67(4):768-73. 2006
- Richards HL, Ray DW, Kirby B, Mason D, Plant D, Main CJ, Fortune DG, Griffiths CE. Response of the hypothalamic-pituitary-adrenal axis to psychological stress in patients with psoriasis. Br J Dermatol. 153(6):1114-20; 2005
- Knoflach M, Kiechl S, Mayr A, Willeit J, Poewe W, Wick G. Allergic rhinitis, asthma, and atherosclerosis in the Bruneck and ARMY studies. Arch Intern Med. 165(21):2521-6; 2005.
- Cutolo M, Villaggio B, Otsa K, Aakre O, Sulli A, Seriolo B. Altered circadian rhythms in rheumatoid arthritis patients play a role in the disease's symptoms. Autoimmun Rev. 4(8):497-502. 2005
- Wolters M. Diet and psoriasis: experimental data and clinical evidence. Br J Dermatol. 153(4):706-14; 2005
- Lukaczer D, Darland G, Tripp M, Liska D, Lerman RH, Schiltz B, Bland JS. A pilot trial evaluating Meta050, a proprietary combination of reduced iso-alpha acids, rosemary extract and oleanolic acid in patients with arthritis and fibromyalgia. Phytother Res.19(10):864-9; 2005
- Eijsbouts AM, van den Hoogen FH, Laan RF, Hermus AR, Sweep CG, van de Putte LB. Hypothalamic-pituitary-adrenal axis activity in patients with rheumatoid arthritis. Clin Exp Rheumatol. 23(5):658-64; 2005
- Bos JD, de Rie MA, Teunissen MB, Piskin G. Psoriasis: dysregulation of innate immunity. Br J Dermatol. 152(6):1098-107; 2005
- Yahiro K, Shirasaka D, Tagashira M, Wada A, Morinaga N, Kuroda F, Choi O, Inoue M, Aoyama N, Ikeda M, Hirayama T, Moss J, Noda M. Inhibitory effects of polyphenols on gastric injury by Helicobacter pylori VacA toxin. Helicobacter. 10(3):231-9; 2005
- Messmer EM. Ocular allergies. Ophthalmologe. 102(5):527-43; 2005
- Goronzy JJ, Weyand CM. Rheumatoid arthritis. Immunol Rev.204:55-73; 2005
- Denburg JA, Hatfield HM, Cyr MM, Hayes L, Holt PG, Sehmi R, Dunstan JA, Prescott SL. Fish oil supplementation in pregnancy modifies neonatal progenitors at birth in infants at risk of atopy. Pediatr Res. 57(2):276-81; 2005
- Groneberg DA, Bester C, Grutzkau A, Serowka F, Fischer A, Henz BM, Welker P. Mast cells and vasculature in atopic dermatitis--potential stimulus of neoangiogenesis. Allergy. 60(1):90-7; 2005
- McBeth J, Chiu YH, Silman AJ, Ray D, Morriss R, Dickens C, Gupta A, Macfarlane GJ. Hypothalamic-pituitary-adrenal stress axis function and the relationship with chronic widespread pain and its antecedents. Arthritis Res Ther. 7(5):R992-R1000; 2005
- Mori TA, Beilin LJ. Omega-3 fatty acids and inflammation, Curr Atheroscler Rep;6(6):461-7; 2004
- Crofford LJ, Young EA, Engleberg NC, Korszun A, Brucksch CB, McClure LA, Brown MB, Demitrack MA. Basal circadian and pulsatile ACTH and cortisol secretion in patients with fibromyalgia and/or chronic fatigue syndrome. Brain Behav Immun. 18(4):314-25; 2004
- Barden AE, Mori TA, Dunstan JA, Taylor AL, Thornton CA, Croft KD, Beilin LJ, Prescott SL. Fish oil supplementation in pregnancy lowers F2-isoprostanes in neonates at high risk of atopy.Free Radic Res. 38(3):233-9; 2004
- Raap U, Werfel T, Jaeger B, Schmid-Ott G. Atopic dermatitis and psychological stress. Hautarzt. 54(10):925-9; 2003
- Buske-Kirschbaum A, von Auer K, Krieger S, Weis S, Rauh W, Hellhammer D. Blunted cortisol responses to psychosocial stress in asthmatic children: a general feature of atopic disease?. Psychosom Med.65(5):806-10; 2003
- Buske-Kirschbaum A, Hellhammer DH. Endocrine and immune responses to stress in chronic inflammatory skin disorders. Ann N Y Acad Sci.992:231-40; 2003
- Cutolo M, Sulli A, Pizzorni C, Craviotto C, Straub RH. Hypothalamic-pituitary-adrenocortical and gonadal functions in rheumatoid arthritis. Ann N Y Acad Sci.992:107-17; 2003
- Ataoglu S, Ozcetin A, Yildiz O, Ataoglu A. Evaluation of dexamethasone suppression test in fibromyalgia patients with or without depression. Swiss Med Wkly. 133(15-16):241-4; 2003
- Eichenfield LF, Hanifin JM, Beck LA, Lemanske RF Jr, Sampson HA, Weiss ST, Leung DY. Atopic dermatitis and asthma: parallels in the evolution of treatment. Pediatrics.111(3):608-16; 2003
- Harle P, Cutolo M, Scholmerich J, Straub RH. Rheumatoid arthritis - pathogenetic role of neuroendocrine axes and the peripheral nervous system. Med Klin (Munich). 97(12):720-9; 2002
- Simopoulos AP. Omega-3 fatty acids in inflammation and autoimmune diseases. J Am Coll Nutr. 21(6):495-505; 2002
- Gil A. Polyunsaturated fatty acids and inflammatory diseases. Biomed Pharmacother. 56(8):388-96; 2002
- Tsigos C, Chrousos GP. Hypothalamic-pituitary-adrenal axis, neuroendocrine factors and stress. J Psychosom Res. 53(4):865-71; 2002
- Harbuz M. Neuroendocrine function and chronic inflammatory stress. Exp Physiol. 87(5):519-25; 2002
- Bennett RM. Adult growth hormone deficiency in patients with fibromyalgia. Curr Rheumatol Rep. 4(4):306-12; 2002
- Okifuji A, Turk DC. Stress and psychophysiological dysregulation in patients with fibromyalgia syndrome. Appl Psychophysiol Biofeedback. 27(2):129-41; 2002
- Mayser P, Mayer K, Mahloudjian M, Benzing S, Kramer HJ, Schill WB, Seeger W, Grimminger F. A double-blind, randomized, placebo-controlled trial of n-3 versus n-6 fatty acid-based lipid infusion in atopic dermatitis. JPEN J Parenter Enteral Nutr.;26(3):151-8; 2002
- Geenen R, Jacobs JW, Bijlsma JW. Evaluation and management of endocrine dysfunction in fibromyalgia. Rheum Dis Clin North Am. 28(2):389-404; 2002
- Cutolo M, Straub RH, Foppiani L, Prete C, Pulsatelli L, Sulli A, Boiardi L, Macchioni P, Giusti M, Pizzorni C, Seriolo B, Salvarani C. Adrenal gland hypofunction in active polymyalgia rheumatica. effect of glucocorticoid treatment on adrenal hormones and interleukin 6. J Rheumatol. 29(4):748-56; 2002
- Mayser P, Grimm H, Grimminger F. n-3 fatty acids in psoriasis. Br J Nutr. 87 Suppl 1:S77-82; 2002
- Cutolo M, Foppiani L, Minuto F. Hypothalamic-pituitary-adrenal axis impairment in the pathogenesis of rheumatoid arthritis and polymyalgia rheumatica. J Endocrinol Invest. 25(10 Suppl):19-23; 2002
- Parker AJ, Wessely S, Cleare AJ. The neuroendocrinology of chronic fatigue syndrome and fibromyalgia. Psychol Med.31(8):1331-45; 2001
- Morand EF, Leech M. Hypothalamic-pituitary-adrenal axis regulation of inflammation in rheumatoid arthritis. Immunol Cell Biol.;79(4):395-9; 2001
- Furse RK, Rossetti RG, Zurier RB. Gammalinolenic acid, an unsaturated fatty acid with anti-inflammatory properties, blocks amplification of IL-1 beta production by human monocytes. J Immunol.167(1):490-6; 2001
- Dekkers JC, Geenen R, Godaert GL, Glaudemans KA, Lafeber FP, van Doornen LJ, Bijlsma JW. Experimentally challenged reactivity of the hypothalamic pituitary adrenal axis in patients with recently diagnosed rheumatoid arthritis. J Rheumatol. 28(7):1496-504; 2001
- Neeck G, Crofford LJ. Neuroendocrine perturbations in fibromyalgia and chronic fatigue syndrome. Rheum Dis Clin North Am. 26(4):989-1002; 2000
- Straub RH, Gluck T, Cutolo M, Georgi J, Helmke K, Scholmerich J, Vaith P, Lang B. The adrenal steroid status in relation to inflammatory cytokines (interleukin-6 and tumour necrosis factor) in polymyalgia rheumatica. Rheumatology (Oxford). 39(6):624-3; 2000
- Torpy DJ, Papanicolaou DA, Lotsikas AJ, Wilder RL, Chrousos GP, Pillemer SR. Responses of the sympathetic nervous system and the hypothalamic-pituitary-adrenal axis to interleukin-6: a pilot study in fibromyalgia. Arthritis Rheum. 43(4):872-80; 2000
- Mastorakos G, Ilias I. Relationship between interleukin-6 (IL-6) and hypothalamic-pituitary-adrenal axis hormones in rheumatoid arthritis.Z Rheumatol. 59 Suppl 2:II/75-9; 2000
- Eijsbouts AM, Murphy EP. The role of the hypothalamic-pituitary-adrenal axis in rheumatoid arthritis. Baillieres Best Pract Res Clin Rheumatol. 13(4):599-613; 1999
- Winfield JB. Pain in fibromyalgia. Rheum Dis Clin North Am. 25(1):55-79; 1999
- Demir H, Kelestimur F, Tunc M, Kirnap M, Ozugul Y. Hypothalamo-pituitary-adrenal axis and growth hormone axis in patients with rheumatoid arthritis. Scand J Rheumatol. 28(1):41-6; 1999
- Buske-Kirschbaum A, Jobst S, Hellhammer DH. Altered reactivity of the hypothalamus-pituitary-adrenal axis in patients with atopic dermatitis: pathologic factor or symptom?.Ann N Y Acad Sci. 840:747-54; 1998
- Demitrack MA, Crofford LJ. Evidence for and pathophysiologic implications of hypothalamic-pituitary-adrenal axis dysregulation in fibromyalgia and chronic fatigue syndrome. Ann N Y Acad Sci.840:684-97; 1998
- Crofford LJ. The hypothalamic-pituitary-adrenal stress axis in fibromyalgia and chronic fatigue syndrome. Z Rheumatol. 57 Suppl 2:67-71; 1998
- Jongen-Lavrencic M, Peeters HR, Wognum A, Vreugdenhil G, Breedveld FC, Swaak AJ. Elevated levels of inflammatory cytokines in bone marrow of patients with rheumatoid arthritis and anemia of chronic disease. J Rheumatol. 24(8):1504-9; 1997
- Buske-Kirschbaum A, Jobst S, Psych D, Wustmans A, Kirschbaum C, Rauh W, Hellhammer D, Attenuated free cortisol response to psychosocial stress in children with atopic dermatitis. Psychosom Med. 59(4):419-26; 1997
- Di Girolamo N, Visvanathan K, Lloyd A, Wakefield D. Expression of TNF-alpha by human plasma cells in chronic inflammation. J Leukoc Biol. 61(6):667-78; 1997
- Calder PC, n-3 polyunsaturated fatty acids and cytokine production in health and disease. Ann Nutr Metab. 41(4):203-34; 1997
- Crofford LJ, Engleberg NC, Demitrack MA. Neurohormonal perturbations in fibromyalgia. Baillieres Clin Rheumatol. 10(2):365-78; 1996
- Nishioka T, Kurokawa H, Takao T, Kumon Y, Nishiya K, Hashimoto K. Differential changes of corticotropin releasing hormone (CRH) concentrations in plasma and synovial fluids of patients with rheumatoid arthritis (RA). Endocr J. 43(2):241-7. 1996
- I. Kyrou, A. Christou, D. Panagiotakos, C.Stefanaki, K.Skenderi, K. Katsana, C. Tsigos. Effects of a hops (Humulus lupulus L.) dry extract supplement on self- reported depression, anxiety and stress levels in apparently healthy young adults: a randomized, placebo-controlled, double-blind, crossover pilot study. Hormones. 16(2):171-180. 2017
- Wall R, Ross RP, Fitzgerald. Fatty acids from fish: the anti-inflammatory potential of long-chain omega-3 fatty acids. Nutr Rev;68(5):280-9. 2010